New tools revolutionise bacteria research
Researchers have generated new standards and tools for research into bacteria. One consequence is greater understanding of how bacteria adapt to humans, so we are better able to develop medicines for combating bacteria that cause diseases.
A new study describes with an extremely high degree of detail how the Bacillus subtilis bacterium adapts to its surroundings.
The result has been achieved using an innovative set of tools that has raised the standard for future bacteria research.
“It’s not so much the results of our research that are interesting as the methods we’ve used to get these results,” says Simon Rasmussen, an assistant professor at the Technical University of Denmark’s Department of Systems Biology.
Together with Hanne Østergaard Jarmer, an associate professor in the same department, Rasmussen was responsible for the Danish contribution to new research in the EU-supported BaSysBio project.
Studied everything bacteria-related
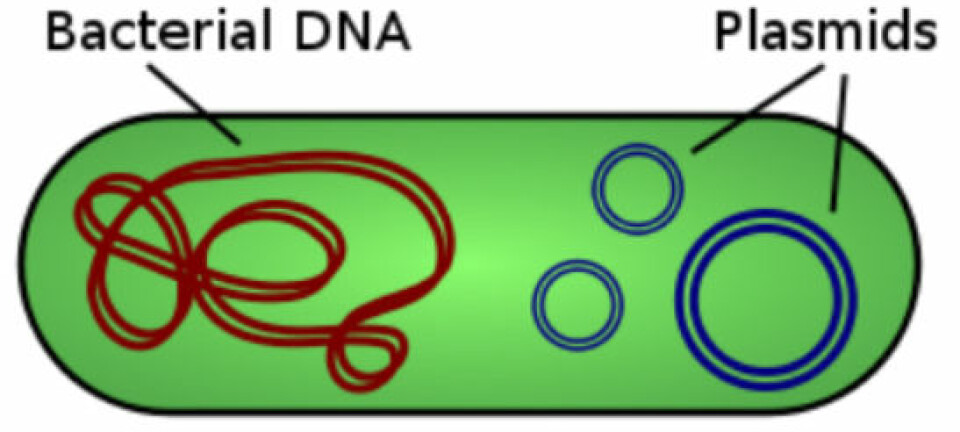
The Bacillus subtilis bacterium is used traditionally as a model organism for Gram-positive bacteria.
Used by industrial companies to produce enzymes, it is also closely related to the anthrax bacterium, Bacillus anthracis.
The international research team wanted to generate a holistic picture of all the processes in Bacillus subtilis when it adapts to a new environment. Therefore they studied how the bacterium reacted when they moved it from one source of energy and gave it a choice of two energy sources.
The choice made by the bacterium is expressed as the initiation of many complex processes in the cell, and it is these processes that the researchers have now mapped in great detail.
“Systems biology, which is our field of work, is about looking at all the processes involved in a biological system, rather than just a few individual elements,” says Rasmussen. While researchers up to now have looked at a small number of genes, for the BaSysBio project the Danish researchers looked at them all.
“Our newly developed methods can be used to look at how all forms of bacteria adapt to a given environment,” adds Rasmussen.
“This applies not only when they adapt to energy sources, but also to the human immune system, which identifies and combats bacteria that cause diseases. With a greater understanding of how bacteria adapt to humans, we will be better able in the future to develop medicines for combating bacteria that cause diseases.”
Bacteria adapt genes to new situation
A bacterium that is adapting to a new energy source or environment must also adapt the genes it uses.
When the researchers changed the bacterium’s energy source from glucose to malate, or from malate to glucose, the expression of up to 2,000 genes was controlled precisely in the bacterium’s cell.
The gene expression is controlled by the bacterium regulating various promoters, which control the levels of transcripts. (See Factbox)
In order to form the overall picture, the international research team had to map the way the bacterium managed its various cell promoters, transcripts, metabolites and proteins.
“In itself, having to choose between malate and glucose is quite simple,” says Rasmussen.
“But measuring all the processes involved in the bacterium’s choice meant we had to develop completely new techniques and mathematical models. Here we discovered that the processes involved in changing from glucose to malate are very different from those used in changing from malate to glucose.”
Rasmussen and Jarmer contributed to the research by designing and optimising some genetic chips used to study the activity of the transcripts.
Economic gains
The new tools can pave the way for economic gains. They can make it easier for industry to better optimise the growth of bacteria for e.g. enzyme production – a growth increase of one percent can give producers of enzymes for washing powders large gains.
Other research projects have been started with the help of the new tools developed during the BaSysBio project.
Researchers have used these new tools for in-depth studies of the feared bacterium MRSA (multidrug- or methicillin-resistant Staphylococcus aureus), which is especially troublesome in hospitals and nursing homes.
The anthrax bacterium, Bacillus anthracis, is also being studied further using the new tools.
These bacteria have not yet been studied as thoroughly or in the same way as Bacillus subtilis, but in the longer term greater knowledge of the functions of bacteria can lead to effective treatment of the diseases they cause.
“Our objective with this research was to make tools that other researchers can use to gain better knowledge of bacteria,” says Rasmussen.
“Our research was therefore on an extremely high level in terms of data precision. We have also published our results on a website and made them more accessible and more easily understood for others.”
Translated by: Michael de Laine
Scientific links
- Global Network Reorganization During Dynamic Adaptations of Bacillus subtilis Metabolism, Science: Vol. 335 no. 6072 pp. 1099-1103 DOI: 10.1126/science.1206871
- Condition-Dependent Transcriptome Reveals High-Level Regulatory Architecture in Bacillus subtilis, Science: Vol. 335 no. 6072 pp. 1103-1106 DOI: 10.1126/science.1206848