How basic research helps fine-tune cancer treatments
Scientists have discovered a surprising mechanism that controls cell-repairing proteins. The new research could substantially improve cancer treatments in the future.
This article is part of our Basic Research theme
Scientists have discovered a hitherto unknown mechanism in the body’s fight against erroneously dividing cells. In the long run, it could help doctors to fine-tune cancer treatments that are currently undergoing clinical trials.
The discovery was made in ATR proteins created inside our cells.
“The discovery could help to improve cancer treatments, which are now being tested and have shown promising results,” says lead-scientist Niels Mailand, professor from the Novo Nordisk Foundation Center for Protein Research, at the University of Copenhagen, Denmark.
Mailand and his team recently published their results in the scientific journal, Nature Cell Biology.
A sensitive process
The discovery is the result of basic research into the way in which the body treats damaged DNA, the so-called DNA damage response.
When cells grow and divide, they form copies of themselves, including their DNA.
“It’s a sensitive process. There’s many things that can stress a cell. For example, if the cells lose their building-blocks, or there is damaged DNA in the copied cell,” says Mailand.
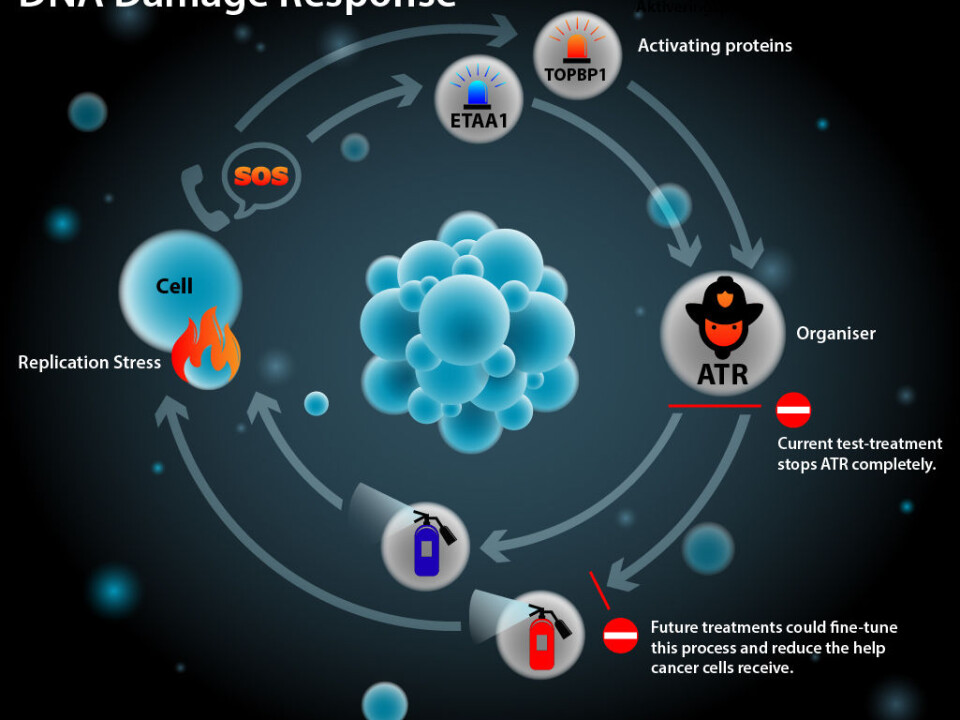
This can trigger a process known as DNA replication stress, which increases the risk of faulty sequences in the copied DNA. This can happen in any cell in the body.
Replication stress activates a protein called ATR to ease the stress and help the cell replication and division process continue as normal.
“The ATR proteins are the cells’ organiser in the fight against replication stress. Until now, we thought that ATR could only be activated by the TopBP1 protein, but we’ve now discovered that another protein can release ATR,” says Mailand.
Read More: Scientists: we will never find a cure for cancer
Cancer unravels the body’s ability to repair cells
Diseases like cancer depend on the body’s ability to fight replication stress.
“Cancer cells grow very quickly, and some grow too quickly for their own good. This means there’s a high chance of mistakes,” says Mailand.
And cancer cells make a lot of mistakes during the replication process so they are very dependent on the ATR-protein to ease the process, he says.
“You could say that cancer cells high jack the body’s ability to repair itself,” says Mailand.
New treatments, currently being tested, use this knowledge to inhibit ATR and prevent cancers from growing too quickly. Unfortunately, this approach also targets healthy cells.
Read More: Recycling approved drugs for cancer treatment
Fine-tune the fight against cancer
It’s here that the new discovery can play an important role.
“Until now we thought that it was a certain protein that initiated ATR, but we’ve shown that the ETAA1 protein also activates the ATR protein’s fight against replication stress,” says Mailand.
Slowing down ATR could help in the fight against cancer. Today, doctors can completely switch off ATR, but the new discovery can fine-tune the way in which doctors attack cancer by closing off specific parts of ATR.
“It can be advantageous as it’s not merely an effective way to halt ATR’s function. Shutting off individual ETAA1 or TopB1 can perhaps be a more gentle [treatment] for [the] patient,” says Mailand.
Read More: Vitamin C supplements increase effectiveness of cancer treatment
ETAA1 plays an important role in cancer cell defence
Mailand is certain that ETAA1 could be an important factor in cancer cells’ stress relief.
Analysis show that some cancer cells some have very little ETAA1, while other have a lot.
“That tells me that ETAA1 plays an important role in the cancer cells’ defence against replication stress,” he says.
Other researchers agree.
“This is absolutely important and exciting work,” says Lise Lotte Hansen, a lecturer at the Department of Biomedicine at Aarhus University, Denmark. She was not involved in the new research but has read the study.
“Mailand and his group have discovered what I would describe as a “missing link” in identifying the function of ETAA1. We’ve tried lots of ways to slow down ATR, but this discovery can create a better way to attack cancer,” says Hansen.
She agrees that the discovery can lead to improved cancer treatments in the future.
“It looks like shutting down ETAA1 will attack cancer cells while leaving healthy cells alone. This could be a tremendous addition to cancer treatments because this replication mechanism is so universally important. This mechanism occurs in all cells and also in all types of cancer. It will be an enormous breakthrough if we can hit, and only hit, cancer cells.”
Read More: Is immunotherapy really a revolution?
Scientists will find cancer’s Achilles’ heel
The next step for Mailand and his team is to figure out precisely what ETAA1 and TopBP1 trigger in the ATR protein.
“We’ve discovered how ETAA1 activates ATR at the molecular level. The next step is to determine exactly how and when ETAA1 protein is important for the cells,” says Mailand.
“That means finding the Achilles’ heel that allows us to hit the cancer cells harder than other cells. It could be that cancer cells need ETAA1 more than healthy cells,” he says.
-------------
Read the Danish version of this article on Videnskab.dk
Translated by: Catherine Jex